Year 11 Atomic Structure Rubric
A free science worksheet ready for your classroom. Open in Kuraplan to grab the print-ready PDF, customize it for your students, or generate a fresh version in seconds.

Year 11 Atomic Structure Rubric
Assessment Criteria & Mark Allocation (Total: 20 marks)
This rubric outlines the assessment criteria and mark allocation for the Year 11 Science assessment on atomic structure and bonding.
Marking Schedule Breakdown
• 4 questions × 1 mark each
• Topics: Atomic number, mass number, basic bonding identification
• Mark allocation: 1 mark for correct answer, 0 marks for incorrect
• Correct Answers:
- 1. Atomic Number: Number of protons in an atom.
- 2. Mass Number: Total number of protons and neutrons in an atom.
- 3. Ionic Bonding: Transfer of electrons between atoms.
- 4. Covalent Bonding: Sharing of electrons between atoms.
• Define atomic number (1 mark), mass number (1 mark), ionic bonding (1 mark), covalent bonding (1 mark)
• Mark allocation: 1 mark for accurate definition including key scientific terms
• 0.5 marks for partially correct definition missing key elements
• Correct Answers:
- Atomic Number: The number of protons in the nucleus of an atom, which determines the element's identity.
- Mass Number: The sum of protons and neutrons in the nucleus of an atom.
- Ionic Bonding: A type of chemical bond formed through the electrostatic attraction between oppositely charged ions.
- Covalent Bonding: A type of chemical bond where two atoms share one or more pairs of electrons.
• Draw Bohr model for given element (e.g., Carbon-12)
• Mark allocation:
- Correct nucleus representation (1 mark)
- Accurate electron shells/energy levels (2 marks)
- Correct electron distribution (2 marks)
- Proper labelling of components (1 mark)
• Correct Answer:
- Bohr model for Carbon-12: 6 protons and 6 neutrons in the nucleus, 2 electrons in the first shell, and 4 electrons in the second shell.
• Explain formation of one ionic and one covalent compound
• Mark allocation:
- Ionic bonding explanation (3 marks): electron transfer (1), charge formation (1), attraction forces (1)
- Covalent bonding explanation (3 marks): electron sharing (1), bond formation (1), molecular structure (1)
• Correct Answers:
- Ionic Bonding Example: Sodium Chloride (NaCl) - Sodium (Na) loses one electron to become Na+, and Chlorine (Cl) gains that electron to become Cl-, resulting in an ionic bond due to the electrostatic attraction between Na+ and Cl-.
- Covalent Bonding Example: Water (H2O) - Two hydrogen atoms share electrons with one oxygen atom, forming covalent bonds and creating a bent molecular structure.
Assessment Guidelines
• Award marks for scientifically accurate content even if expressed in student's own words
• Deduct 0.5 marks for significant spelling errors in scientific terminology
• Partial marks available for incomplete but correct working in diagram questions
• No penalty for minor drawing imperfections if scientific accuracy maintained
Excellence (18-20 marks): Comprehensive understanding, accurate application, excellent diagrams
Merit (14-17 marks): Good understanding with minor gaps, mostly accurate work
Achieved (10-13 marks): Basic understanding, some correct applications, adequate diagrams
Not Achieved (0-9 marks): Limited understanding, significant errors, poor diagram quality
About This Worksheet
Free in Kuraplan
Sign up free, grab the PDF, and customize it for your class.
Print-Ready
Formatted for standard paper. Clean layout, easy to read.
AI-Generated
Created with Kuraplan's AI, designed for real classroom use.
For Teachers & Parents
Use in classrooms, for homework, tutoring, or homeschool.
Need a custom version of this worksheet?
Kuraplan's AI generates custom worksheets in seconds — differentiated for every learner, aligned to your curriculum.
Generate Custom Worksheets — FreeMore Science Worksheets

Variables in Science Investigations
Free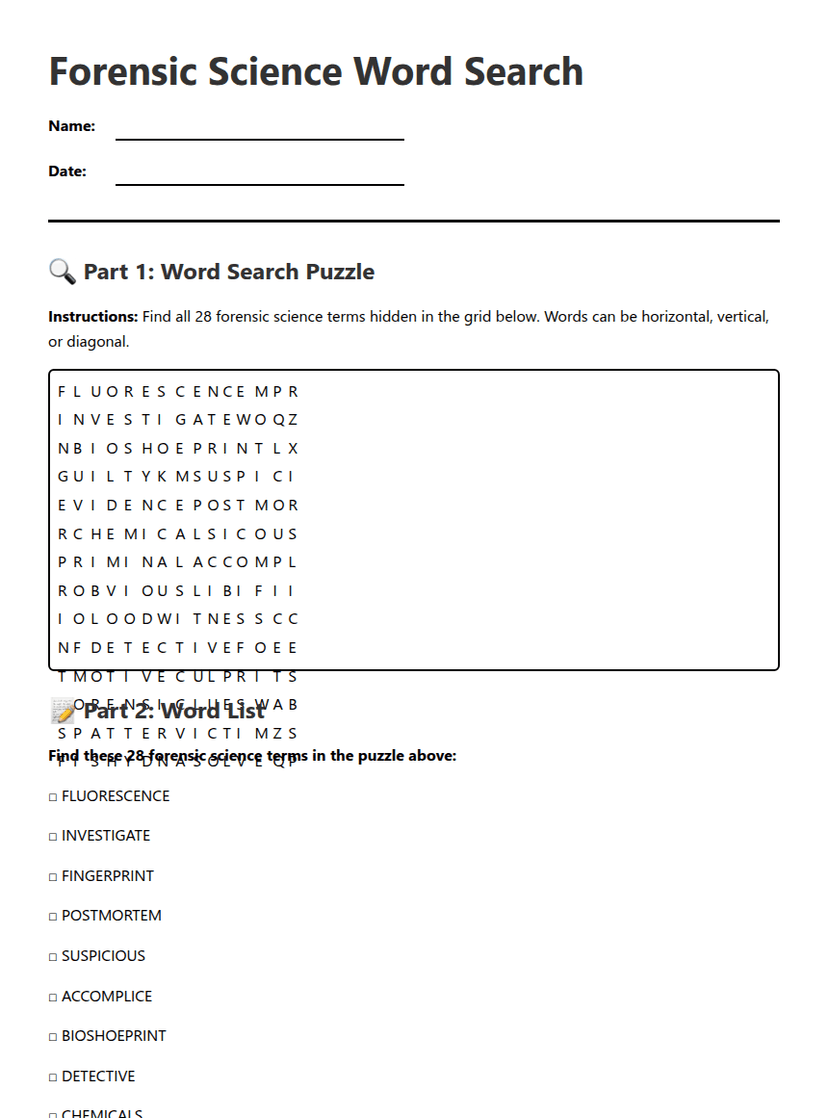
Forensic Science Word Search
Free
Science Observation Recording Sheet
Free
Science Objects Exploration
Free
Sports Science Introduction
Free
Senior Social Science Icebreaker
Free
What is Science?
Free