Matter & Decimals Practice
A free science worksheet ready for your classroom. Open in Kuraplan to grab the print-ready PDF, customize it for your students, or generate a fresh version in seconds.

Matter & Decimals Practice
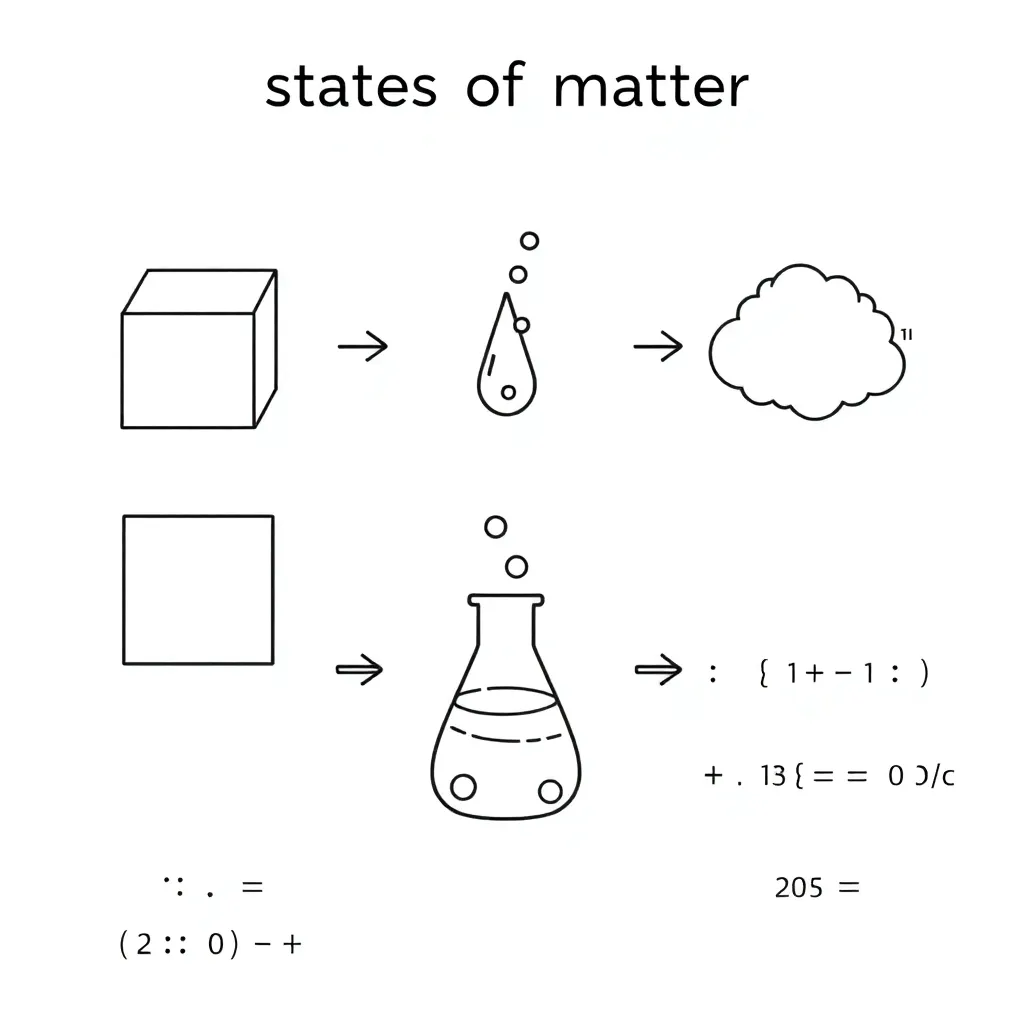
🔬 Part 1: Adding Decimals with Particle Quantities
I can: Add decimals using partial sums and explain that matter is made of particles.
⚗️ Part 2: Subtracting Decimals with Physical & Chemical Changes
I can: Subtract decimals using the difference method and identify physical and chemical changes.
🧪 Part 3: Mixed Practice Word Problems
I can: Solve real-world problems combining matter concepts with decimal operations.
About This Worksheet
Free in Kuraplan
Sign up free, grab the PDF, and customize it for your class.
Print-Ready
Formatted for standard paper. Clean layout, easy to read.
AI-Generated
Created with Kuraplan's AI, designed for real classroom use.
For Teachers & Parents
Use in classrooms, for homework, tutoring, or homeschool.
Need a custom version of this worksheet?
Kuraplan's AI generates custom worksheets in seconds — differentiated for every learner, aligned to your curriculum.
Generate Custom Worksheets — FreeMore Science Worksheets

Variables in Science Investigations
Free
Forensic Science Word Search
Free
Science Observation Recording Sheet
Free
Science Objects Exploration
Free
Sports Science Introduction
Free
Senior Social Science Icebreaker
Free
What is Science?
Free