Chemical Bonding Formulas
A free, printable science worksheet ready for your classroom. Download instantly, print, and hand out to your students — no account needed.

Chemical Bonding Formulas
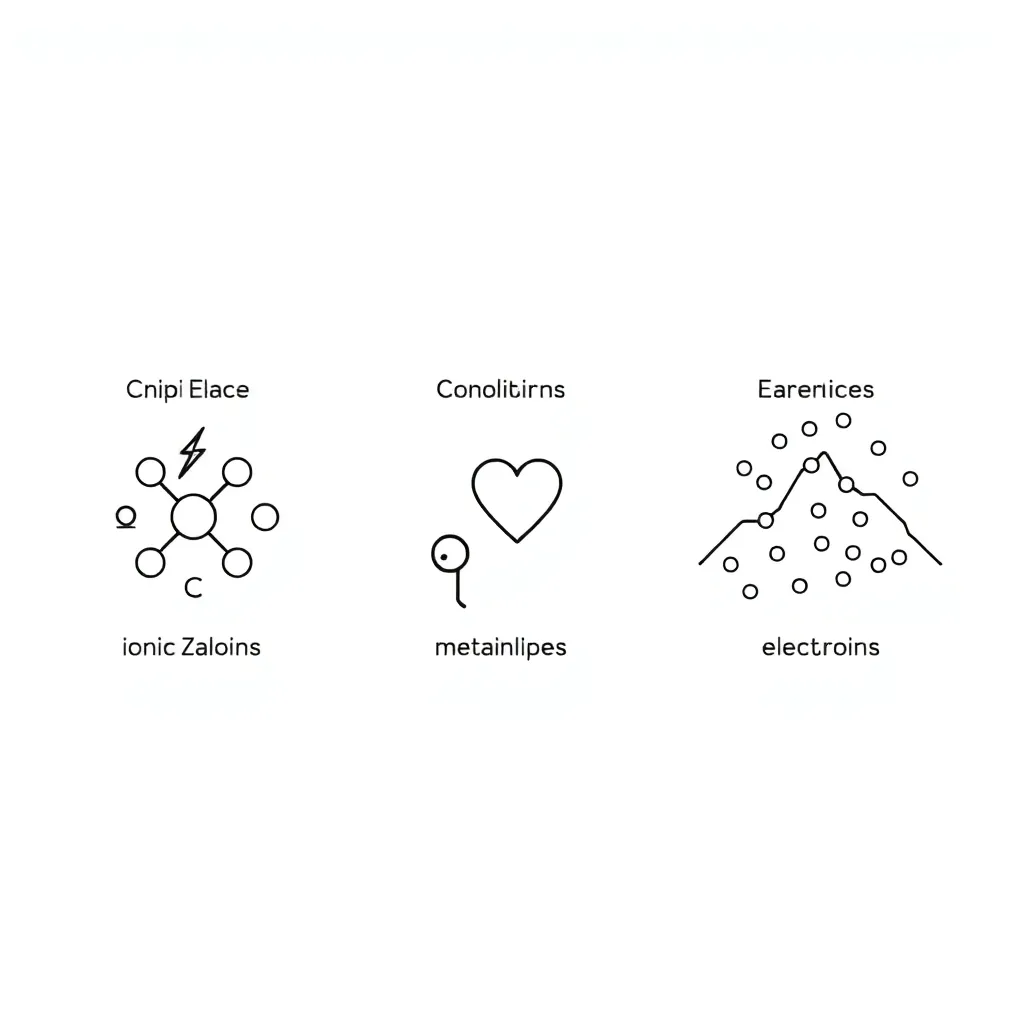
🔬 Part 1: Understanding Chemical Bonds
Ionic Bonds: Form when electrons are transferred from metal atoms to non-metal atoms. The metal becomes a positive ion and the non-metal becomes a negative ion. These opposite charges attract each other. Example: Sodium chloride (NaCl) - table salt.
Covalent Bonds: Form when non-metal atoms share electrons with each other. The shared electrons hold the atoms together. Example: Water (H₂O) - hydrogen and oxygen share electrons.
Metallic Bonds: Form between metal atoms where electrons move freely in a "sea of electrons" around the metal atoms. This gives metals their special properties like conductivity. Example: Aluminium foil (Al).
🎯 Part 2: Matching Chemical Compounds to Bond Types
⚗️ Part 3: Writing Chemical Formulas
Common Elements and Their Charges:
Metals: Na⁺, Mg²⁺, Al³⁺, K⁺, Ca²⁺
Non-metals: Cl⁻, O²⁻, F⁻, S²⁻, N³⁻
Covalent elements: H, C, N, O, F, Cl
a) Sodium chloride: ____________
b) Magnesium oxide: ____________
c) Calcium fluoride: ____________
d) Aluminium oxide: ____________
a) Water (hydrogen + oxygen): ____________
b) Carbon dioxide (carbon + oxygen): ____________
c) Methane (carbon + hydrogen): ____________
d) Ammonia (nitrogen + hydrogen): ____________
🧪 Part 4: Apply Your Knowledge
Chemical bonds form when atoms share or transfer ______________. The type of bond affects the ______________ of the compound. ______________ bonds form between metals and non-metals, ______________ bonds form between non-metals, and ______________ bonds form between metals.
About This Worksheet
Free Download
No sign-up, no email, no paywall. Just download and print.
Print-Ready
Formatted for standard paper. Clean layout, easy to read.
AI-Generated
Created with Kuraplan's AI, designed for real classroom use.
For Teachers & Parents
Use in classrooms, for homework, tutoring, or homeschool.
Need a custom version of this worksheet?
Kuraplan's AI generates custom worksheets in seconds — differentiated for every learner, aligned to your curriculum.
Generate Custom Worksheets — Free

