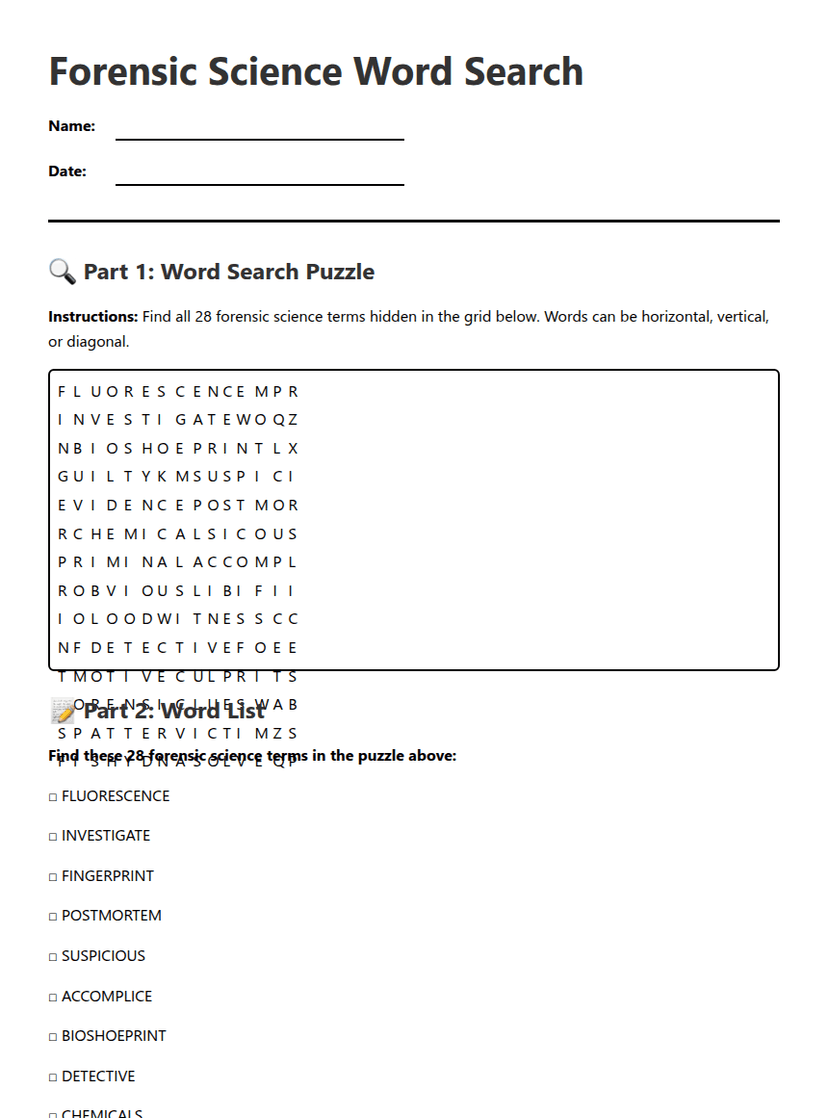
Year 11 Atomic Structure Rubric
A free, printable science worksheet ready for your classroom. Download instantly, print, and hand out to your students — no account needed.

Year 11 Atomic Structure Rubric
Assessment Criteria & Mark Allocation (Total: 20 marks)
This rubric outlines the assessment criteria and mark allocation for the Year 11 Science assessment on atomic structure and bonding.
Marking Schedule Breakdown
• 4 questions × 1 mark each
• Topics: Atomic number, mass number, basic bonding identification
• Mark allocation: 1 mark for correct answer, 0 marks for incorrect
• Correct Answers:
- 1. Atomic Number: Number of protons in an atom.
- 2. Mass Number: Total number of protons and neutrons in an atom.
- 3. Ionic Bonding: Transfer of electrons between atoms.
- 4. Covalent Bonding: Sharing of electrons between atoms.
• Define atomic number (1 mark), mass number (1 mark), ionic bonding (1 mark), covalent bonding (1 mark)
• Mark allocation: 1 mark for accurate definition including key scientific terms
• 0.5 marks for partially correct definition missing key elements
• Correct Answers:
- Atomic Number: The number of protons in the nucleus of an atom, which determines the element's identity.
- Mass Number: The sum of protons and neutrons in the nucleus of an atom.
- Ionic Bonding: A type of chemical bond formed through the electrostatic attraction between oppositely charged ions.
- Covalent Bonding: A type of chemical bond where two atoms share one or more pairs of electrons.
• Draw Bohr model for given element (e.g., Carbon-12)
• Mark allocation:
- Correct nucleus representation (1 mark)
- Accurate electron shells/energy levels (2 marks)
- Correct electron distribution (2 marks)
- Proper labelling of components (1 mark)
• Correct Answer:
- Bohr model for Carbon-12: 6 protons and 6 neutrons in the nucleus, 2 electrons in the first shell, and 4 electrons in the second shell.
• Explain formation of one ionic and one covalent compound
• Mark allocation:
- Ionic bonding explanation (3 marks): electron transfer (1), charge formation (1), attraction forces (1)
- Covalent bonding explanation (3 marks): electron sharing (1), bond formation (1), molecular structure (1)
• Correct Answers:
- Ionic Bonding Example: Sodium Chloride (NaCl) - Sodium (Na) loses one electron to become Na+, and Chlorine (Cl) gains that electron to become Cl-, resulting in an ionic bond due to the electrostatic attraction between Na+ and Cl-.
- Covalent Bonding Example: Water (H2O) - Two hydrogen atoms share electrons with one oxygen atom, forming covalent bonds and creating a bent molecular structure.
Assessment Guidelines
• Award marks for scientifically accurate content even if expressed in student's own words
• Deduct 0.5 marks for significant spelling errors in scientific terminology
• Partial marks available for incomplete but correct working in diagram questions
• No penalty for minor drawing imperfections if scientific accuracy maintained
Excellence (18-20 marks): Comprehensive understanding, accurate application, excellent diagrams
Merit (14-17 marks): Good understanding with minor gaps, mostly accurate work
Achieved (10-13 marks): Basic understanding, some correct applications, adequate diagrams
Not Achieved (0-9 marks): Limited understanding, significant errors, poor diagram quality
About This Worksheet
Free Download
No sign-up, no email, no paywall. Just download and print.
Print-Ready
Formatted for standard paper. Clean layout, easy to read.
AI-Generated
Created with Kuraplan's AI, designed for real classroom use.
For Teachers & Parents
Use in classrooms, for homework, tutoring, or homeschool.
Need a custom version of this worksheet?
Kuraplan's AI generates custom worksheets in seconds — differentiated for every learner, aligned to your curriculum.
Generate Custom Worksheets — Free